Non-Human Primate (NHP)
Accelerate Clinical Success with Premier Non-Human Primate (NHP) Research
Bridging the Gap from Bench to Bedside with AAALAC-Accredited Excellence
Prisys Biotech is a leading contract research organization (CRO) dedicated to advancing preclinical and translational medicine through non-human primate (NHP)-based research. Recognized as an AAALAC-accredited facility and a high-tech enterprise in Shanghai, we combine ethical responsibility, scientific excellence, and technological innovation to provide data that truly bridges bench to bedside.
Why Partner with Prisys?
- Global Standards: Fully AAALAC-accredited facility ensuring top-tier animal welfare.
- Clinical-Grade Platform: Equipped with human-clinical operating rooms, MRI, CT, and DSA imaging.
- Specialized Expertise: A dedicated focus on NHP models that rodent studies cannot replace.
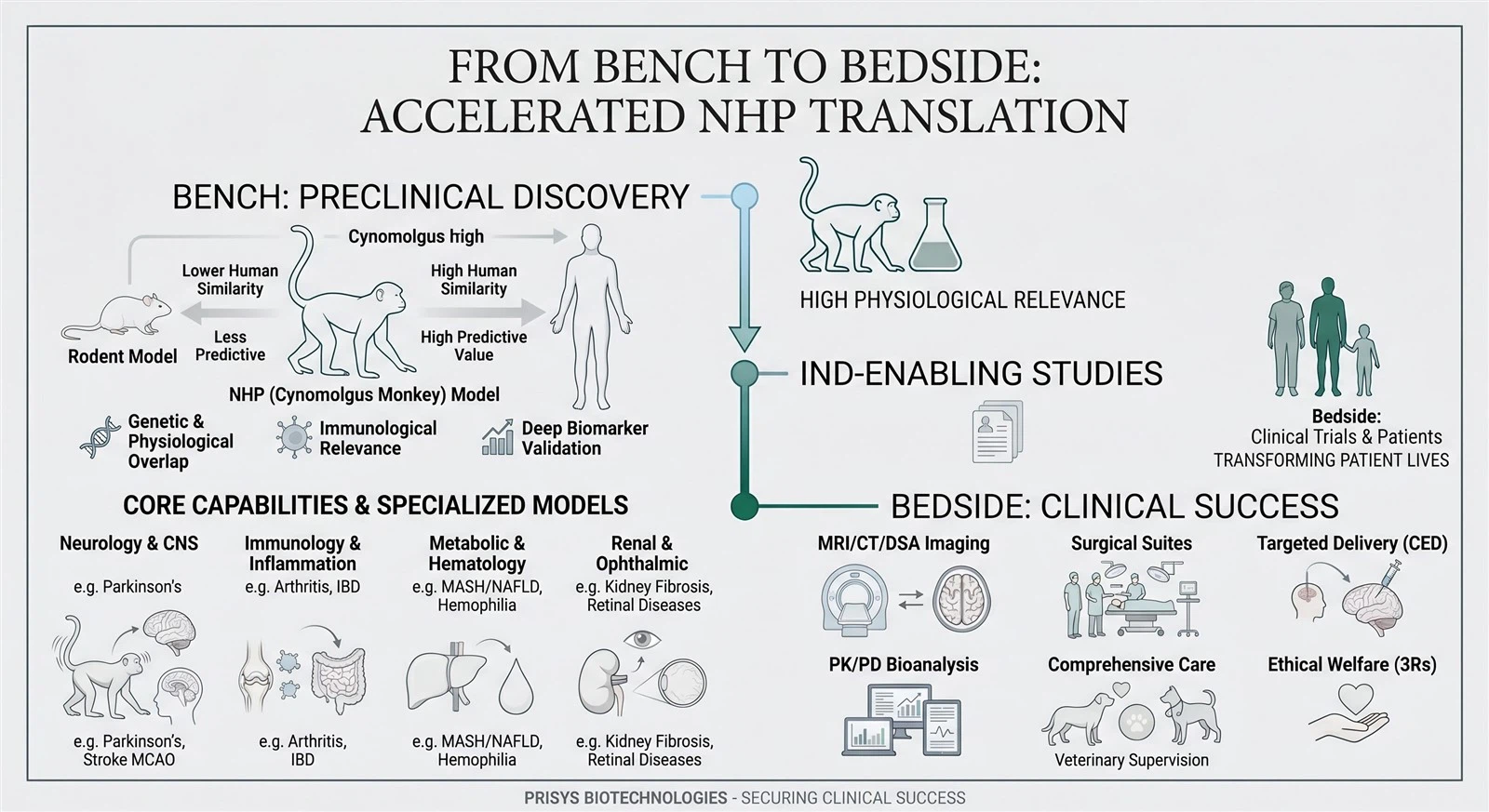
The NHP Advantage: Unmatched Translational Value
Rodent models often fail to capture the complexity of human disease. Our Cynomolgus monkey models share critical genetic, physiological, and immunological similarities with humans, providing the most predictive data for:
-
Neurology & CNS: Parkinson's, Stroke (MCAO), Alzheimer's, Pain.
-
Metabolic & Hematology: MASH/NAFLD, Diabetes, Hemophilia, Thrombosis.
-
Renal & Ophthalmic: Kidney fibrosis, Glaucoma, Retinal diseases.
"We don't just provide data; we provide the confidence to move into clinical trials."
Our Core Capabilities
| Service Domain | Advanced Solutions at Prisys |
| Pharmacology & PD | 40+ Validated Disease Models across 10+ therapeutic areas with deep biomarker validation. |
| Pharmacokinetics (PK) | Precise sampling, bioanalysis, and tissue distribution using clinical-standard protocols. |
| Safety Assessment | Non-GLP/GLP-like toxicology, including CNS, cardiovascular, and respiratory safety pharmacology. |
| Targeted Delivery | MRI-Guided Convection-Enhanced Delivery (CED), Nose-to-Brain (N2B), and DSA-guided organ perfusion. |
| Advanced Imaging | In-house 1.5T MRI, CT, DSA, and Ultrasound for longitudinal, non-invasive monitoring. |
👉 Explore Our Full Catalog of NHP Models
Commitment to Ethics & Animal Welfare
Animal welfare is the foundation of our science.
We operate under strict AAALAC guidelines with pathogen-free housing, sterile ICU units, and comprehensive veterinary care. We strictly adhere to the 3Rs (Replacement, Reduction, Refinement), ensuring every study is scientifically justified and ethically sound.
Ready to De-Risk Your Drug Development?

Join pharmaceutical and biotech innovators worldwide who trust Prisys for high-quality, reproducible IND-enabling data.
📩 Contact Our Scientists Today
Discuss your program needs or request a customized study proposal.

Featured Publication & Case Study
In collaboration with our industry partner, Prisys Biotech successfully developed and validated a Unilateral Ureteral Obstruction (UUO) model in Cynomolgus monkeys. This study addresses a critical gap in translational medicine, providing a reliable platform for testing next-generation therapeutics.
This validated Cynomolgus UUO model is now available at Prisys to test human-specific anti-fibrotic therapeutics with higher translational confidence.
Hot Tags: non-human primate (nhp), research, study, monkey, gene therapy, NHP NASH Model, protein PK pharmacology, Neuropathic Pain Nonhuman Primates, NHP clinical pharmacology, NHP Asthma Dermatitis Model, NHP Biodistribution pharmacology
You Might Also Like
Send Inquiry