This milestone marks a significant step forward in the clinical translation of cell therapies for neurological disorders and highlights the value of coordinated efforts across multiple stakeholders in advancing innovative therapeutics toward the clinic.
Advancing Mechanism-Driven Approaches for Epilepsy Treatment
Epilepsy is a complex and prevalent central nervous system (CNS/Neurology) disorder, with a substantial proportion of patients showing limited response to existing pharmacological treatments. As a result, there remains a clear unmet medical need.
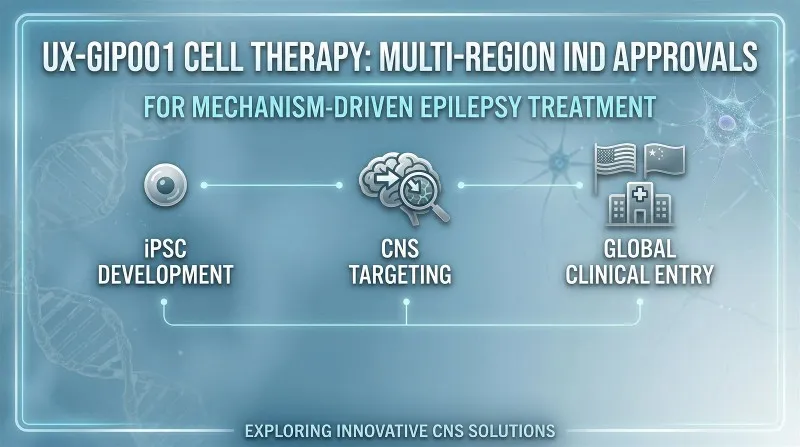
In recent years, cell therapy-based approaches have emerged as a promising strategy in this field. UX-GIP001 is developed based on an induced pluripotent stem cell (iPSC) platform, where human pluripotent stem cells are differentiated in vitro into neural progenitor cells with defined functional characteristics. This approach is designed to modulate abnormal neural circuits associated with epilepsy, providing a mechanism-driven pathway for therapeutic intervention.
During the advancement of this program, Prisys Biotech contributed to the nonclinical stage by supporting intracerebral delivery studies, leveraging its experience in non-human primate (NHP) models and CNS-targeted administration technologies.

Prisys Biotech's Non-Human Primate (NHP) Research Capabilities
Comprehensive NHP Disease Model Portfolio
Non-human primates (NHPs) share high similarity with humans in genetics, physiology, and immune systems, making them highly relevant for Translational-Research-Center. Prisys Biotech has established over 40 disease models with strong clinical relevance, including:
- Neurological disorders: Brain Ischemic Stroke/MCAO, Parkinson's disease, acute and chronic Pain Models
- Immunology and inflammation: Rheumatoid Arthritis/CIA, Atopic dermatitis, Asthma, Psoriasis, Inflammatory Bowel Disease
- Metabolic and renal diseases: Renal Fibrosis/UUO, NASH / NAFLD, Type 2 diabetes, obesity
- Respiratory diseases: idiopathic pulmonary fibrosis (IPF), acute lung inflammation
- Hematology and cardiovascular diseases: bleeding/anemia models, thrombosis (A-V Shunt), Acute Myocardial Infarction
Core Technology Platforms for Translational Research
To bridge the gap between preclinical studies and clinical applications, Prisys Biotech has developed a series of proprietary platforms with strong translational value.
iMRI-Guided Intracerebral Delivery Platform in NHPs
Prisys has established a real-time, MRI-Guided Drug Delivery For CNS Research in NHPs. Supported by the ClearPoint® neuro-navigation system, this platform enables submillimeter trajectory planning and precise targeting of brain regions.
In this study, Convection-Enhanced Delivery (CED) Technology was applied, allowing continuous low-rate, positive-pressure infusion to generate a local convective field within brain tissue. This facilitates volumetric distribution of therapeutics, improving both coverage and homogeneity at the target site.
Combined with intraoperative MRI, the system enables real-time visualization and verification of infusion location and distribution, enhancing procedural control and reproducibility.
Clinical-Grade Multimodal Imaging Platform
Prisys operates a Clinical Imaging center led by experienced radiologists from top-tier hospitals. Equipped with MRI, CT, and digital subtraction angiography (DSA), the platform supports advanced imaging protocols for quantitative analysis of drug distribution and Pharmacokinetics (PK), as well as indirect assessment of target engagement through imaging biomarkers.
AI-Based Markerless 3D Behavioral Analysis System (BehaviorAtlas®)
Prisys has developed an AI-based NHP Behavior Analysis System for NHPs. This platform enables long-term, non-invasive monitoring and quantitative classification of motor dysfunction, gait abnormalities, and pain-related behaviors, providing multidimensional efficacy data for neurological disease studies such as Parkinson's disease and pain models.
About UniXell Biotechnology
UniXell Biotechnology, founded in 2021, focuses on the development of iPSC-based cell therapy products, with a primary emphasis on neurological diseases. The company is actively advancing its pipeline toward clinical translation and exploring innovative therapeutic strategies.
About Prisys Biotech
Prisys Biotech is dedicated to providing non-human primate (NHP) models and translational research services for innovative drug development. The company focuses on NHP Neuroscience research and advanced imaging platforms, integrating imaging-guided technologies with in vivo capabilities to support the nonclinical evaluation of complex biologics, including cell and gene therapies.
FAQ
Q: What is the significance of the UX-GIP001 IND approval?
A: The IND approval for UX-GIP001 from both the FDA and CDE marks a major milestone for UniXell Biotechnology, allowing their iPSC-derived cell therapy for epilepsy to enter clinical studies in both the US and China.
Q: How did Prisys Biotech support the UX-GIP001 program?
A: Prisys Biotech supported the nonclinical development by providing expertise in NHP models and performing MRI-guided intracerebral delivery studies using Convection-Enhanced Delivery (CED) to ensure precise drug distribution.
Q: What unique technologies does Prisys Biotech offer for CNS research?
A: Prisys offers an iMRI-guided stereotactic delivery platform, CED technology for volumetric distribution, a clinical-grade multimodal imaging center, and an AI-driven 3D behavioral analysis system (BehaviorAtlas®).