NHP Intrathecal (IT) Injection & CSF Sampling Services | Prisys Biotech
Technical Overview: Intrathecal (IT) Administration and CSF Sampling in NHPs
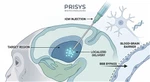
In the development of CNS-targeted therapeutics-such as Antisense Oligonucleotides (ASOs), gene therapies, and biologics-surpassing the blood-brain barrier (BBB) remains a critical challenge. Prisys Biotech provides standardized, high-precision Intrathecal (IT) Administration and Cerebrospinal Fluid (CSF) Sampling services in Non-Human Primates (NHPs) to support pharmacokinetic (PK) and pharmacodynamic (PD) profiling.
1. Dual-Route Access: Cisterna Magna and Lumbar Puncture
Our platform utilizes two primary anatomical routes to ensure localized drug delivery and systemic monitoring:

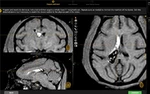
- Ultrasound-Guided Cisterna Magna (CM) Puncture: We employ real-time ultrasonography to visualize the subarachnoid space. This ensures the needle penetrates the dura mater precisely, minimizing the risk of trauma to the brainstem or spinal cord.
- Lumbar Puncture (LP): By optimizing spinal flexion, our clinicians maximize the intervertebral space for reliable lumbar access. This route is typically utilized for bolus dosing or catheter integration.
2. Advanced Catheterization and Chronic Porting
For longitudinal studies requiring frequent sampling or continuous infusion, Prisys offers specialized surgical interventions:
- Subcutaneous IT Porting: Implanted ports allow for repeated access with minimal animal stress. Ports are maintained with heparinized saline to ensure long-term patency.
- Indwelling Catheters: Precision placement of catheters (with beveled ends directed caudally) enables continuous CSF collection.
- CSF Volume Management: To maintain physiological intracranial pressure, we strictly regulate collection volumes (typically <2ml/day) and utilize artificial CSF (aCSF) supplementation where necessary.
3. Quality Control and Analytical Validation
The integrity of CNS data depends on the purity of the collected biofluids. Prisys adheres to rigorous internal benchmarks:
- Contamination Monitoring: Every sample is inspected for blood contamination. Hemolyzed samples are documented and flagged to prevent interference with LC-MS/MS or ELISA-based assays.
- Standardized Processing: We provide integrated analytical support, including high-sensitivity LC-MS/MS analysis for test articles (TA) to generate accurate CSF concentration-time curves.
- Verification of Success: Correct placement is consistently verified by the presence of clear, spontaneous CSF flow prior to any dosing or sampling event.

4. Applications in NHP Research
- Pharmacokinetics: Determination of drug half-life and distribution within the CNS compartment.
- Biomarker Discovery: Longitudinal monitoring of neuroinflammatory or neurodegenerative markers in CSF.
- Safety Pharmacology: Assessment of local tissue tolerability and neurological impact of IT-administered compounds.
Technical Specifications Summary
| Feature | Specification / Method |
| Species | Cynomolgus Macaque (Macaca fascicularis), Rhesus Macaque |
| Guidance | Real-time Ultrasound (Cisterna Magna) |
| Dosing Volume | Total ~18ml/animal capacity; controlled daily limits |
| Sampling Frequency | Up to 7+ samples within 8-24 hours (study dependent) |
| Analytical Support | Integrated LC-MS/MS, Protein Analysis |
Hot Tags: NHP Intrathecal (IT) Injection & CSF Sampling Services | Prisys Biotech, research, study, monkey, gene therapy, ADME, adme of drug, Non human primate PK, Pharmacokinetics, pk and pd, PK PD ADME
You Might Also Like
Send Inquiry