Intracerebral injection (commonly abbreviated as ICM, though other abbreviations exist) is a direct administration route involving the injection of a therapeutic agent directly into specific, targeted regions of the brain parenchyma, such as the hippocampus, striatum, cortex, basal ganglia, or thalamus. This technique is frequently employed in neuroscience research and therapeutic development, utilizing stereotaxic instruments and fine needles to achieve precise intervention within the desired brain area.
1. Precise Localization of the Administration Site
Accurate targeting is fundamental to ICM administration.
- Non-Clinical Research: The choice of ICM injection site is driven by the scientific objectives of the study. When targeting specific brain regions (e.g., striatum or hippocampus), Magnetic Resonance Imaging (MRI)-compatible stereotaxic frames and atlases are often used to precisely determine coordinates, ensuring the drug is delivered accurately to the intended area.
- Clinical Application: In the clinical setting, where the goal is patient treatment, the injection site selection is tailored to the individual patient's condition, considering factors like the location of the lesion and the specific brain structure, to ensure the therapeutic reaches the target effectively.
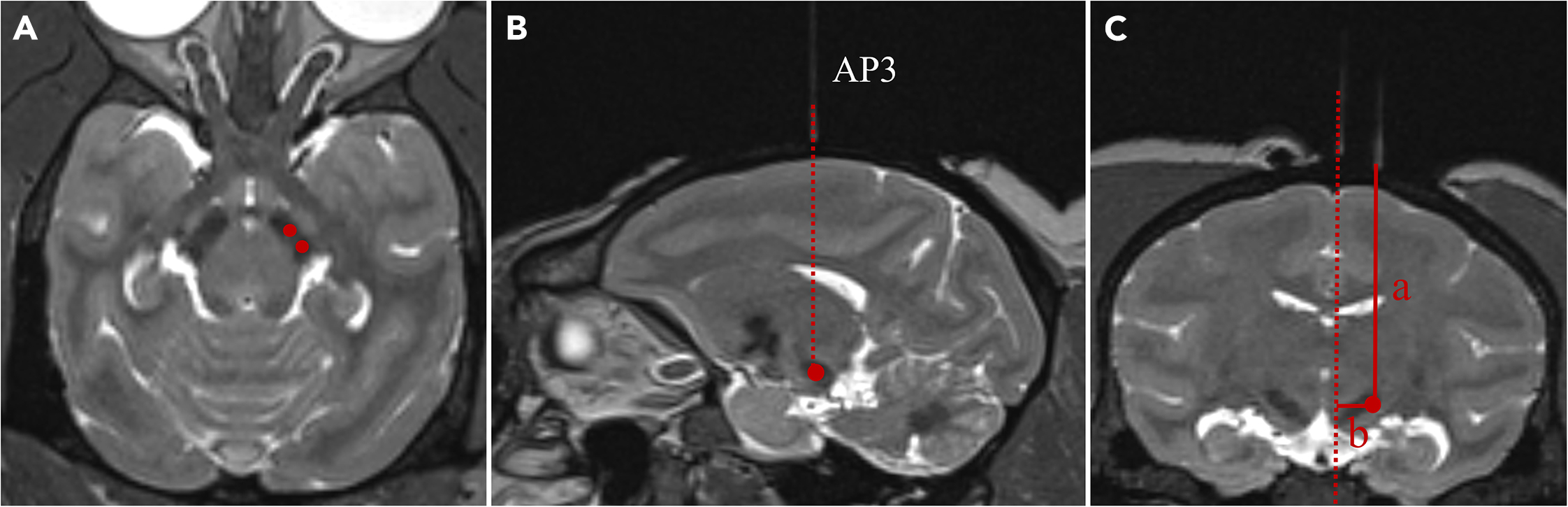
2. Applicability and Suitability
ICM administration is most suitable for therapeutic scenarios requiring rapid achievement of high local drug concentrations within a specific brain region, such as in the treatment of certain brain tumors or potentially focal conditions following stroke.
Contraindications: This route is generally not recommended for patients with elevated intracranial pressure (ICP) or hemorrhagic stroke, as the procedure could exacerbate these conditions. Furthermore, ICM is less suitable for CNS diseases with pathology widely distributed throughout the spinal cord, brainstem, and cortex.
Monitoring & Risk Assessment:
- Clinical: Thorough patient assessment is crucial before the procedure, including ICP measurement and evaluation for hydrocephalus, to prevent complications like infection or cerebral edema. Close post-injection monitoring is essential to manage any adverse events promptly.
- Non-Clinical: Preclinical studies must rigorously evaluate potential procedural risks, including localized hemorrhage, infection, or neural damage at the injection site. Post-administration behavioral assessments (such as AI-based NHP Behavior Analysis System) are also vital to evaluate the drug's impact on CNS function.
3. Advantages and Disadvantages of ICM Administration
Advantages:
- Bypasses the Blood-Brain Barrier (BBB): ICM delivers the drug directly into the brain parenchyma, circumventing the BBB's limitations.
- High Local Concentration: This route significantly increases drug concentration within the targeted brain region, which can enhance therapeutic efficacy, particularly evident in brain tumor treatments where achieving high local drug levels is critical.
- Potentially Superior Efficacy for Localized Conditions: In specific contexts, like certain models of acute ischemic stroke, direct intracerebral administration has demonstrated greater efficacy compared to systemic (e.g., intravenous) routes.
- Optimized Target Exposure: As demonstrated by the gene therapy Upstaza, ICM can achieve significantly higher drug concentrations/expression specifically within the target brain structure (e.g., putamen) compared to other direct CNS routes like intracerebroventricular (ICV) or intrathecal (IT) administration, even at lower doses, highlighting its advantage for highly localized targets.

Disadvantages:
- Invasiveness and Surgical Risk: ICM is an invasive procedure requiring precise stereotaxic surgery performed under strict aseptic conditions. It carries inherent surgical risks.
- Limited Diffusion: Drug distribution may be limited primarily to the area immediately surrounding the injection site, potentially restricting the therapeutic reach if the target area is broad or poorly defined.
- Potential Complications: Risks include infection, hemorrhage, direct brain tissue injury, and cerebral edema. Transient hypothermia and procedural pain can also occur.
- Clinical Compliance: The invasive nature can impact patient acceptance and compliance in clinical settings.
- Non-Clinical Pathology: Common histopathological findings in non-clinical studies include inflammatory reactions, neuronal damage, gliosis, and hemorrhage at or near the injection site.
4. Representative Drug: Upstaza
Upstaza (eladocagene exuparvovec), developed by PTC Therapeutics, is an innovative gene therapy utilizing an Adeno-Associated Virus serotype 2 (AAV2) vector. It is approved for treating Aromatic L-amino Acid Decarboxylase (AADC) deficiency in patients 18 months and older.

Disease Background: AADC deficiency is a rare, autosomal recessive genetic disorder caused by mutations in the DDC gene. This gene is normally expressed in the striatum and encodes the AADC enzyme, crucial for synthesizing key neurotransmitters dopamine and serotonin. Enzyme deficiency leads to severe motor dysfunction, developmental delays, and other neurological symptoms.
Mechanism of Action: Upstaza is administered via MRI-guided stereotactic surgery directly into the putamen, a region of the brain where the DDC gene product is functionally important. The therapy delivers a functional copy of the DDC gene, aiming to restore AADC enzyme production and consequently dopamine synthesis, thereby addressing the underlying genetic defect.
Non-Clinical & Clinical Findings: A pivotal non-clinical study in juvenile non-human primates compared ICM (putamen delivery, 1.2×10¹⁰ vg), ICV (1.2×10¹¹ vg), and IT (1.2×10¹¹ vg) administration. All routes were generally well-tolerated. Notably, despite the ICM dose being ten times lower than the ICV and IT doses, gene expression in the target putamen region was significantly higher following ICM administration. This supported the selection of ICM as the most effective and appropriate route for Upstaza. Clinical data show meaningful improvements in motor and cognitive function observed as early as 3 months post-treatment, with durability sustained for at least 10 years. Commonly reported adverse reactions include initial insomnia, irritability, and dyskinesia; significant procedure-related safety warnings have not been prominently highlighted based on available public information.
Harnessing ICM for Precision Brain Therapy
Intracerebral (ICM) injection provides an unparalleled level of precision for delivering therapeutics directly to targeted regions within the brain, offering a potent way to bypass the BBB and maximize local drug exposure. However, its invasiveness and associated risks demand meticulous planning and execution, particularly during non-clinical development. Comprehensive safety assessment must evaluate procedural safety, local tissue tolerance, potential neurotoxicity, and functional consequences.
Successfully navigating the complexities of ICM administration in non-clinical programs requires specialized expertise in stereotaxic procedures, appropriate animal model selection, and thorough toxicological evaluation. Collaborating with experienced partners, such as Prisys Biotech, can provide crucial support in designing and executing these challenging studies, ultimately facilitating the translation of promising CNS therapies to the clinic.