In the preclinical research phase of innovative drug development, the accurate interpretation of pharmacokinetic (PK) data is pivotal. Non-compartmental Analysis (NCA), as a classic statistical method in pharmacokinetics, has become the preferred tool for regulatory submissions (IND) and academic publications due to its independence from specific compartment assumptions.

Why is NCA the Gold Standard for PK Analysis?
The process of a drug within the body is typically summarized as ADME (Absorption, Distribution, Metabolism, and Excretion). To quantify this process, researchers rely on mathematical models. Compared to complex compartmental models, Non-compartmental Analysis (NCA) utilizes mathematical methods such as the trapezoidal rule to calculate parameters directly from the drug concentration-time data.
Because it requires fewer assumptions and provides robust, reproducible results, NCA is widely applied in preclinical and clinical pharmacology studies for a vast range of therapeutics, from small molecules to large molecule biologics.
Part 01. Detailed Interpretation of Core PK Parameters
Parameters calculated via NCA can be categorized into three main groups: parameters reflecting drug exposure, parameters reflecting time characteristics, and parameters reflecting clearance and distribution properties.
1. Exposure Parameters
These parameters directly reflect the concentration levels of the drug in the body and are critical indicators for evaluating efficacy and safety.
Cmax (Peak Concentration):
Definition: The maximum observed serum/plasma concentration after dosing.
Significance: Closely related to the intensity of the drug's effect and potential toxicity. In multiple-dosing scenarios, it represents the peak concentration within a dosing interval.
Cmin and Ctrough:
Cmin: The minimum concentration observed within a dosing interval.
Ctrough: The concentration immediately prior to the next dose at steady state (end of the dosing interval).
Note: At steady state, if the pre-dose concentration is the lowest point, the numerical values of Cmin and Ctrough may be identical, but their definitions differ (see table below).
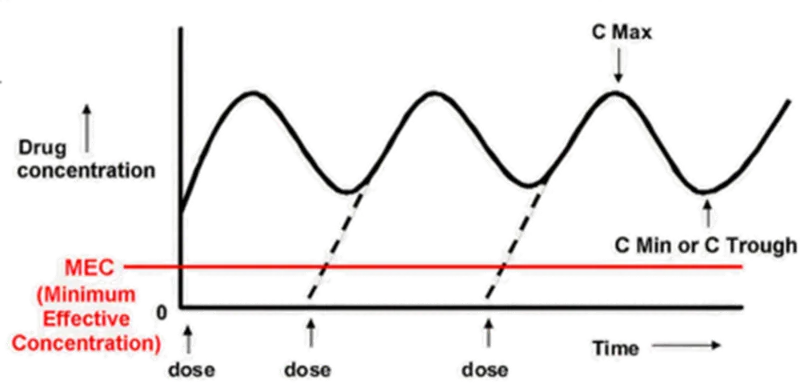
Figure: Steady-state drug concentration-time curve, showing the relationship between fluctuation range and Minimum Effective Concentration - MEC
Cavg (Average Concentration):
Definition: The average concentration during a dosing interval at steady state (Formula: AUCtau / τ).
Significance: For drugs where efficacy is proportional to total exposure (e.g., certain antibiotics or oncology drugs), Cavg is a vital reference indicator.
AUC (Area Under the Curve):
AUClast: The area under the curve from time zero to the last quantifiable time point.
AUCinf: The area under the curve extrapolated from time zero to infinity.
Quality Control: The ratio of AUClast / AUCinf should be ≥ 80%. This ensures that the sampling duration was sufficient to cover the drug's primary elimination phase.
AUCtau: The area under the curve within a dosing interval at steady state.
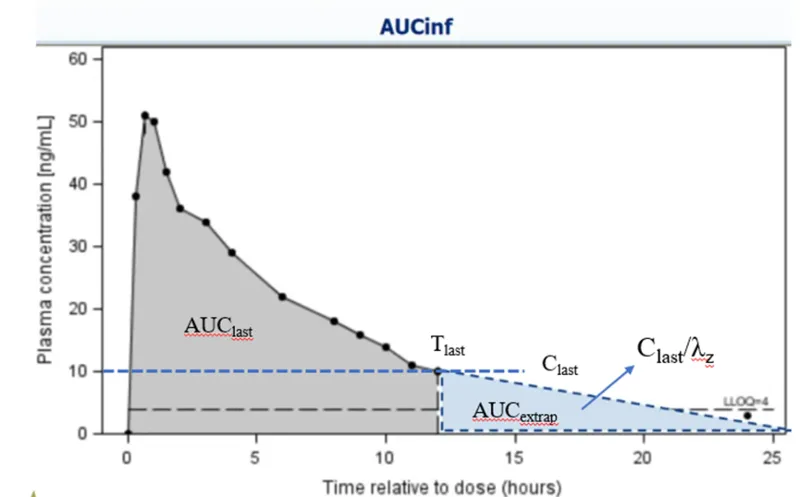
Figure: Calculation and extrapolation of AUC-The shaded area represents AUClast, while the triangular area under the dashed line represents the extrapolated portion
2. Time Parameters
Tmax: The time at which Cmax is observed. For immediate-release formulations, this reflects the absorption rate; for sustained-release formulations, it reflects release characteristics.
Tlag (Lag Time): The time delay between drug administration and the first measurable concentration. It is often used to evaluate the disintegration delay of enteric-coated formulations.
T1/2 (Terminal Elimination Half-life):
Definition: The time required for the drug concentration to decrease by half during the elimination phase (Formula: ln2 / λz).
Significance: Determines the dosing frequency and the time required to reach steady state (typically 5–7 half-lives).
3. Clearance and Distribution Parameters
λz (Lambda z, Terminal Elimination Rate Constant):
Derived from the negative slope of the linear regression of the log-transformed terminal concentration-time data. It is the basis for calculating T1/2 and AUCinf.
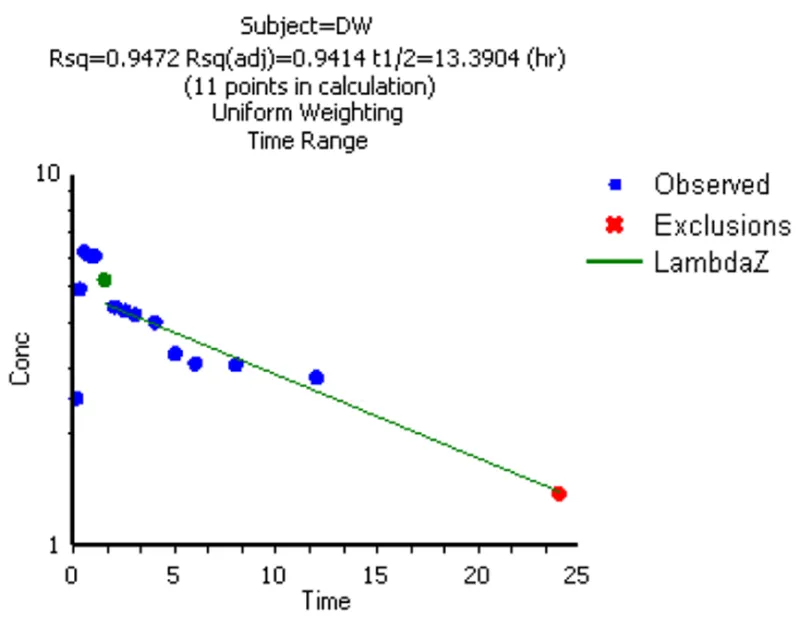
Figure: Linear regression fitting using terminal data points to calculate λz
CL (Clearance):
The volume of plasma from which the drug is completely removed per unit of time.
Intravenous (IV): CL = Dose / AUCinf
Extravascular (e.g., PO, SC): CL/F = Dose / AUCinf (where F is Bioavailability). Since F is often unknown, this calculates the Apparent Clearance.
Vz (Volume of Distribution):
A theoretical volume reflecting the extent of drug distribution in the body.
Calculation: Vz = CL / λz.
Significance: A large Vz indicates the drug is extensively distributed in tissues; a small Vz (close to plasma volume) suggests the drug is confined primarily to the blood.
4. Special Parameters for Multiple Dosing
DF (Fluctuation): Calculated as (Cmax - Ctrough) / Cavg. This reflects the amplitude of concentration fluctuation at steady state; smaller fluctuations generally correlate with a better safety profile.
Rac (Accumulation Ratio): The ratio of steady-state AUC to single-dose AUC. If Rac > 1, it indicates drug accumulation in the body, suggesting a need to adjust the dosing regimen.
Part 02. The Cornerstone of High-Quality PK Research: Precise Animal Experimentation
Accurate PK parameter calculation depends entirely on high-quality raw data. In NCA, the design of sampling time points, the precision of sampling operations, and the standardization of sample processing directly determine the reliability of Cmax, Tmax, and terminal phase fitting.
The Prisys Biotech Advantage
As a clinical preclinical CRO specializing in Non-human Primate (NHP) and other large animal models, Prisys Biotech offers unique advantages in PK/PD research:
- High-Standard Experimental Platform: Our facility is fully AAALAC accredited, ensuring animal welfare and the international acceptance of experimental data.
- Precision Operations Team: Our experienced technical team can execute complex administration routes (e.g., Intracerebral, Intravitreal, Intrathecal injections) and high-frequency precision sampling, ensuring the accurate capture of Tmax.
- Translational Medicine Perspective: Leveraging a rich library of disease models (e.g., hepatic metabolism, cardiovascular, and CNS diseases), we provide scientific interpretation of PK characteristics under pathological conditions, enabling a seamless translation from Pre-clinical to Clinical phases.
Conclusion
Prisys Biotech is committed to being a leader in large animal pharmacology and efficacy research. If you require high-level PK/PD research services, disease model development, or safety evaluation support, please contact us.
Contact Us
Website: www.prisysbiotech.com
Business Email: bd@prisysbiotech.com
Address: Building 35, No. 6055 Jinhai Highway, Fengxian District, Shanghai, China