At Prisys Biotechnology, we acknowledge the growing interest in oligonucleotide-based drugs, particularly small interfering RNA (siRNA) therapeutics. siRNA represents the third generation of therapeutic drugs following small molecule drugs and antibody-based therapies, and has shown great potential in revolutionizing the field of drug development.
siRNA Mechanism of Action:
siRNAs are short nucleotide sequences of about 20 bases that utilize Watson-Crick base pairing principles to bind to complementary mRNA strands, thereby influencing transcription and translation processes. Based on their structural and mechanistic variations, siRNAs are classified as antisense oligonucleotides (ASO), small interfering RNAs (siRNA), small activating RNAs (saRNA), microRNAs (miRNA), and aptamers. Among these, siRNA has gained significant attention due to its promising therapeutic efficacy and technological advancements.
Importance of Chemical Modifications and Delivery Systems:
Unmodified siRNAs have a short half-life and pose a risk of off-target toxicity. Chemical modifications and suitable delivery systems play a crucial role in achieving therapeutic effects. The six approved siRNA drugs mentioned earlier have undergone chemical modifications, such as phosphorothioate backbone modifications and ribose modifications like 2'-O-Methyl (2-OMe), 2'-O-Methoxyethyl (2-MOE), and 2'-Fluoro (2-F). Furthermore, the efficiency of siRNA delivery relies on advanced delivery systems such as LNP and GalNAc conjugates, both of which have demonstrated liver-targeting capabilities.
Preclinical Safety Evaluation and Animal Species Selection:
The preclinical safety evaluation of siRNA drugs involves assessing target-specific and off-target toxicities in animal models. The selection of animal species, including rodents and non-human primates (NHP), depends on the specific characteristics of the siRNA drug. For instance, primate species exhibit high homology with humans in terms of target mRNA sequences and provide a better assessment of siRNA pharmacokinetics and pharmacodynamics. Utilizing non-human primates for safety evaluation has been a common practice, especially for siRNA drugs targeting the liver.

Liver Toxicity and Immune Responses:
The liver is a key organ of concern in siRNA therapeutics. The approved siRNA drugs have demonstrated various characteristics associated with liver-specific delivery, hepatocyte uptake, and potential hepatotoxicity. Histopathological studies have revealed hepatocellular vacuolization, inflammatory infiltration, and mild elevations in liver enzymes such as alanine transaminase (ALT) and aspartate transaminase (AST). Immune responses, including anti-drug antibodies (ADA), cytokine release, complement activation, and immune phenotyping, have also been evaluated and monitored.
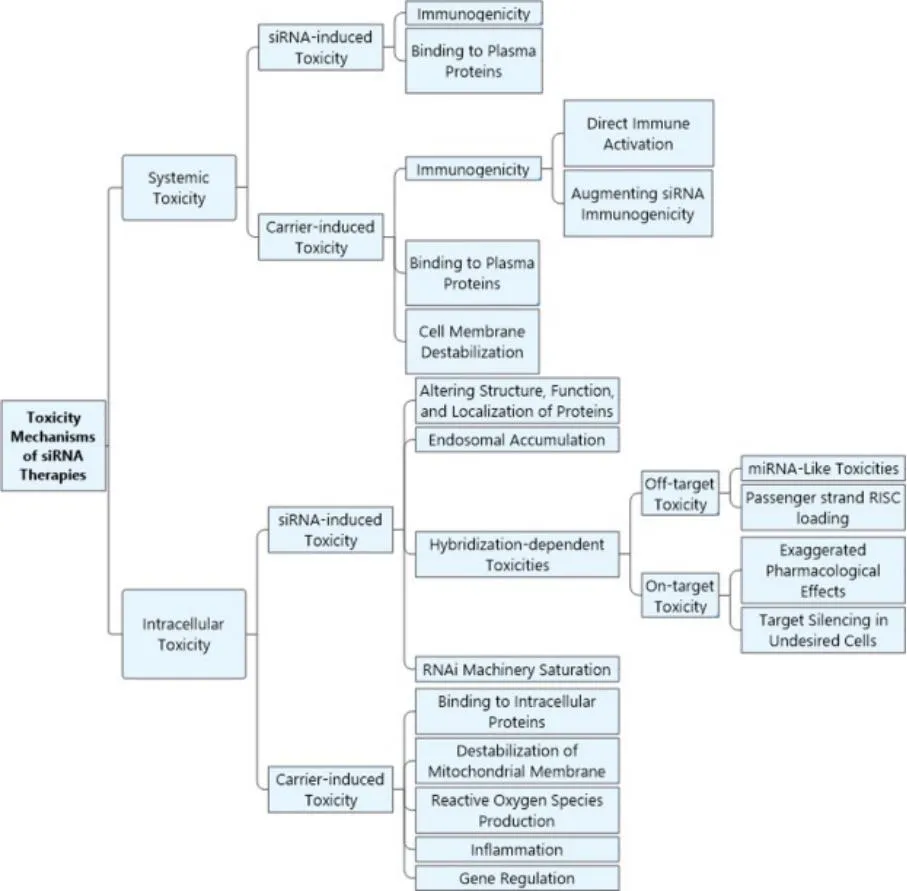
siRNA-mediated gene silencing offers immense therapeutic potential. However, achieving accurate silencing can be challenging. Off-target effects, where siRNA inadvertently suppresses unintended genes, can lead to unforeseen consequences. The Japan Drug and Medical Device Administration (PMDA) recognizes this in its "Guidelines for Non-clinical Safety Evaluation of Oligonucleotide Therapeutic Products," categorizing siRNA toxicity into targeted and off-target types.

Liver: The Prime Target, the Prime Concern
The liver is the primary organ of concern for siRNA drugs. To delve deeper, we analyze six marketed siRNA drugs, focusing on drug characteristics, pharmacokinetics, and observed pathology. Notably, all six target liver-related genes for either synthesis or expression. Additionally, they are chemically modified, like thiophosphate, which can contribute to hepatotoxicity through high protein binding. Both lipid nanoparticles (LNPs) and N-acetylgalactosamine (GalNAc) conjugation facilitate liver targeting. For instance, 80-90% of LNPs in Patisiran, an LNP-siRNA drug, accumulate in the liver upon intravenous administration. GalNAc-siRNAs utilize ASGPR-mediated endocytosis for intracellular uptake, with ASGPR highly expressed in hepatocytes.
Liver Toxicity Profile: From Accumulation to Inflammation
Tissue distribution analyses consistently reveal the highest siRNA concentrations in the liver. Lumasiran, for example, exhibited an AUC of 76700 h*μg/mL and a half-life of 409h in monkeys at 10 mg/kg. Notably, long-term monkey toxicity studies revealed dose-dependent histopathological liver changes:
1. Hepatocyte/Kupffer cell basophilic granules: These might represent the drug, its metabolites, or cellular adaptations to oligonucleotides.
2. Hepatocyte vacuolation: Potentially a secondary effect due to cytoplasmic basophilic granule accumulation.
3. Inflammatory infiltration: High drug concentrations can activate pro-inflammatory pathways.
4. Hepatocyte single-cell necrosis: Severe pathological damage occurs at high doses with extensive basophilic granule accumulation.
Elevated blood biochemistry markers like ALT and AST often accompany these changes. However, comprehensively evaluating tissue damage, associated index alterations, reversibility, and similar findings in other siRNA drugs is crucial for determining the true harmfulness of these changes.
Beyond the Liver: Off-Target Toxicity and Immune Stimulation
Sequence complementarity-independent off-target toxicity also warrants attention. Immune stimulation reactions, largely dependent on nucleotide sequence design and chemical modifications, are one consequence. siRNA drugs can activate the innate immune system, leading to injection site reactions (Patiiran, Inclisiran) and platelet (PLT) decreases (Patisiran, Givosiran). Evaluating immune stimulation involves in vivo analysis of relevant markers and in vitro assessments using isolated peripheral blood mononuclear cells or whole blood to predict cytokine release.
Non-hybridization-related off-target effects, like coagulation function changes, can also occur. Certain siRNA drugs selectively inhibit Factor IXa and cofactor VIIIa in the coagulation cascade, altering thrombin action time and indirectly inhibiting coagulation (e.g., Patisiran, Lumasiran, slightly elevated APTT). Additionally, siRNA drugs can bind to inhibitor H, activating the complement bypass pathway. Overactivation of complement can disrupt its function, leading to secondary inflammation and vasculitis (e.g., Patisiran-induced C3a increase, liver inflammatory infiltration, perivascular inflammation at infusion sites).
Delivery System Toxicity: Don't Overlook the Carrier
The delivery system, often LNPs, deserves attention as well. Novel excipients or altered component ratios in LNPs necessitate thorough investigation. While specific LNP guidelines remain absent, existing guidelines like "Guidance for Industry: Lipiodol Product Chemistry, Manufacturing, and Controls; Human Pharmacokinetics and Bioavailability; And Labeling Documentation" can offer valuable insights. Patisiran's LNP investigations can also serve as a reference. Notably, if LNPs fall under the non-independent excipient registration standard, they can be investigated alongside siRNA with the same animal species as siRNA's non-clinical safety evaluation, considering the two-species requirement for siRNA.

Conclusion:
The field of siRNA therapeutics continues to advance with a focus on improving delivery systems, chemical modifications, minimizing off-target effects, and expanding indications. Prisys Biotechnology remains committed to the development of innovative siRNA-based therapies, leveraging our expertise in drug discovery and delivery systems. We strive to bring forth transformative treatments that address unmet medical needs and improve patient outcomes.