In preclinical bioanalysis, the selection of biological matrix-serum or plasma-is more than a routine decision. It directly impacts data accuracy, assay reliability, and the translatability of pharmacokinetic (PK) and pharmacodynamic (PD) results. At Prisys Biotech, a preclinical CRO specializing in non-human primate (NHP) models and large animal translational research, we emphasize thoughtful matrix selection as a foundational step in early drug development. This article outlines the physiological differences, analytical implications, and strategic considerations behind matrix choice.
Understanding the Biological Matrices
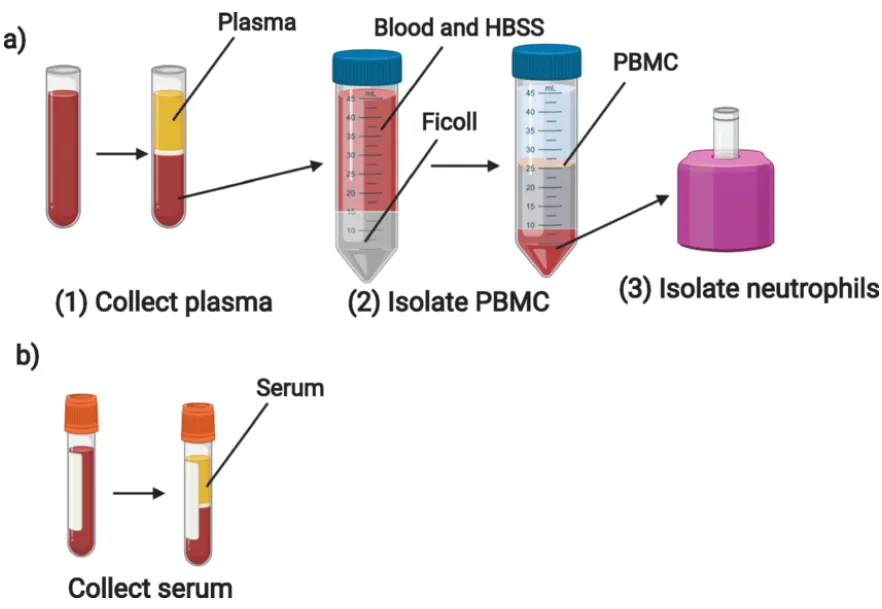
Both serum and plasma are derived from whole blood, but their biochemical compositions differ significantly due to the presence or absence of clotting factors.
Serum is the liquid portion obtained after blood has clotted. During coagulation, fibrinogen converts to fibrin, forming a mesh that traps blood cells. After centrifugation, the remaining fluid-serum-contains proteins, hormones, nucleic acids, and other analytes but lacks fibrinogen and certain clotting factors.
Plasma, on the other hand, is obtained by adding anticoagulants (e.g., EDTA, heparin, citrate) to prevent clotting, followed by centrifugation. Plasma retains fibrinogen and coagulation factors, providing a closer representation of the in vivo physiological state.

Matrix Selection: Why It Matters
Choosing between serum and plasma is not arbitrary. Many analytes exhibit different concentrations depending on the matrix. Mixing matrices within the same study can compromise result integrity, making matrix standardization essential for valid comparisons and regulatory compliance.
Key takeaway: For a given analyte, always use a consistent matrix throughout the study.
Advantages of Plasma
Plasma is increasingly favored in modern bioanalysis, especially for large-animal and NHP studies, due to the following operational and scientific advantages:
1. Faster Sample Processing
Plasma can be centrifuged immediately after collection. In contrast, serum requires a 30-minute clotting period, which can be longer in patients with coagulopathies or under anticoagulant therapy. Incomplete clotting may result in microclots that interfere with downstream assays or instrumentation.
2. Higher Yield
Plasma typically accounts for ~55% of whole blood volume, while serum yields are ~15–20% lower. In studies where sample volume is limited-such as in pediatric or hemophilia models-plasma allows for greater analytical flexibility.
3. Reduced Interference from Clotting
The clotting process alters analyte levels by triggering cellular metabolism and releasing intracellular contents. For example, potassium, LDH, and AST concentrations are often artificially elevated in serum. Conversely, analytes like glucose or platelets may decrease during clotting, making plasma a more stable choice for certain targets.
4. Lower Risk of Assay Disruption
Serum samples often contain fibrin strands or microclots that may block analytical instruments or interfere with immunoassays. Plasma avoids this issue entirely when properly processed.
Advantages of Serum
Despite the benefits of plasma, serum remains a standard matrix in many established protocols and has its own advantages:
1. Lower Risk of Cellular Contamination
Serum samples undergo a full clotting process, effectively removing most cellular elements. In contrast, plasma may retain platelets or cellular fragments if centrifugation is suboptimal, which could degrade sample integrity over time or with freeze-thaw cycles.
2. Stability of Specific Analytes
Certain analytes demonstrate greater chemical stability in serum than plasma. For example, glucose may be more stable in serum under ambient conditions, though this is analyte-specific.
3. No Interference from Anticoagulants
Plasma preparation requires anticoagulants, some of which (e.g., EDTA, heparin) may interfere with downstream detection methods, particularly immunoassays. Serum avoids this risk entirely.
4. Ease of Cross-Study Comparison
Due to its historical prevalence, many reference ranges, standard methods, and legacy datasets are based on serum. For certain programs, this facilitates benchmarking and regulatory submission.
Choosing the Right Matrix: Strategic Considerations
There is no universal answer. Matrix choice must align with:
- Target analyte properties
- Analytical method requirements
- Study timelines and sample handling constraints
- Volume limitations (especially in NHPs or disease models)
- Regulatory or historical precedents
Key recommendation: Do not mix matrices within the same project. Once chosen, use the same matrix consistently to avoid variability and ensure data comparability.
Matrix selection is a critical yet often overlooked decision in preclinical bioanalysis. At Prisys Biotech, we help drug developers make evidence-based choices that ensure analytical consistency and translational success. Whether using plasma to reflect in vivo physiology or serum to align with legacy datasets, we tailor our bioanalytical strategies to the specific needs of each study.
About Prisys Biotech
Prisys Biotech is a preclinical CRO specializing in large-animal pharmacology and translational research, with a focus on non-human primates (cynomolgus monkeys). Our integrated bioanalytical services are designed to support high-quality PK, PD, and toxicology studies for IND-enabling and early-phase development.