Therapeutic proteins, including monoclonal antibodies, have revolutionized the treatment of numerous diseases. However, the development of these powerful drugs is often complicated by immunogenicity-the tendency of a therapeutic to provoke an unwanted immune response in the host. This response can manifest in several ways, from infusion reactions and cytokine release syndrome (CRS) to the formation of Anti-Drug Antibodies (ADAs).
The development of ADAs is a primary concern in immunogenicity assessment. These antibodies can directly neutralize a drug's therapeutic effect by blocking its active site or indirectly impact efficacy by accelerating its clearance from the body. Understanding, predicting, and managing immunogenicity is therefore a critical step in ensuring the safety and success of any biologic drug program.
The Mechanism of ADA Formation: A Multi-Step Process
The generation of ADAs is a complex immunological cascade involving both the innate and adaptive immune systems. The process typically begins with Antigen-Presenting Cells (APCs), such as dendritic cells (DCs).
- Uptake and Processing: After systemic administration (e.g., intravenous or subcutaneous), the biologic drug is encountered and internalized by APCs. Inside the APC, the protein drug is broken down into smaller peptide fragments.
- Antigen Presentation: These peptide fragments (T-cell epitopes) are presented on the surface of the APC by Major Histocompatibility Complex Class II (MHC-II) molecules.
- T-Cell Activation: The peptide-MHC-II complex is specifically recognized by corresponding T-cell receptors (TCRs) on CD4+ helper T-cells. This recognition activates the helper T-cells.
- B-Cell Activation and Antibody Production: Activated helper T-cells then provide signals to specific B-cells, stimulating them to differentiate into plasma cells and produce ADAs.
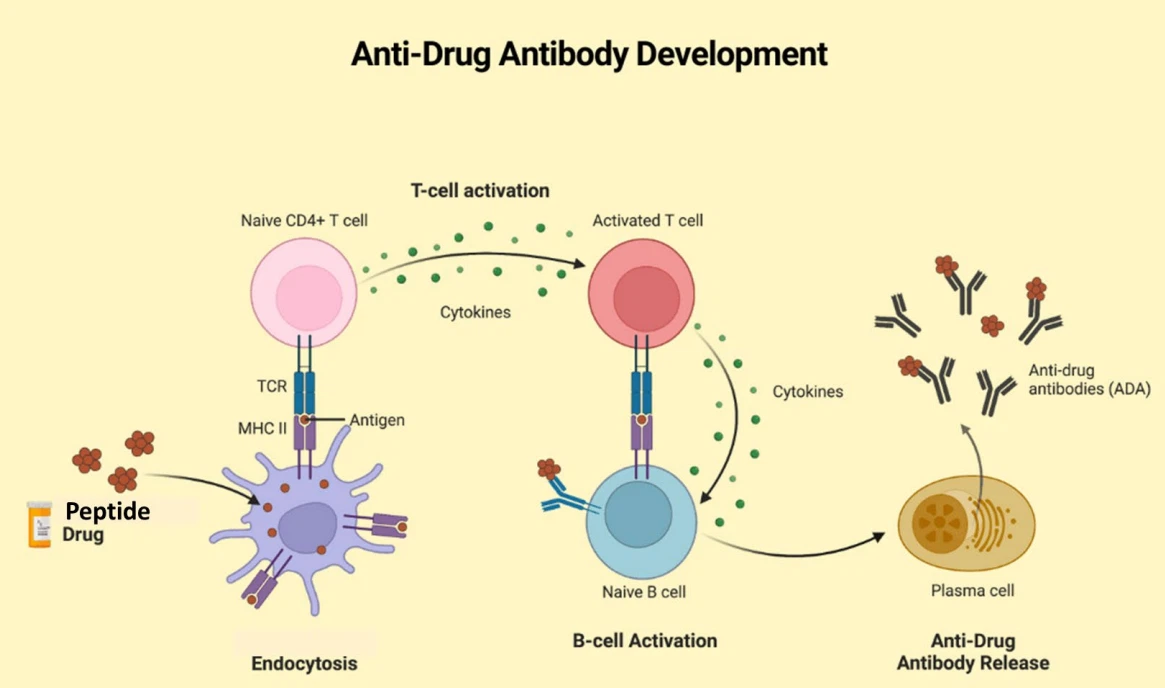
Initially, the ADAs produced are often low-affinity IgM antibodies. Through processes of antibody class-switching and affinity maturation (somatic hypermutation), higher-affinity IgG, IgE, or IgA antibodies can be generated, potentially leading to a more robust and sustained immune response.
Key Risk Factors Influencing Immunogenicity
The risk of ADA formation is influenced by a combination of factors related to the product, the patient, and the disease state. For drug developers, understanding product-related risks is paramount.
Product-Related Factors:
- Molecular Characteristics: The amino acid sequence is a primary driver. Non-human sequences, such as those in chimeric antibodies or fusion proteins, inherently increase immunogenicity risk.
- Formulation and Purity: Product impurities, aggregates, and certain excipients (e.g., some surfactants) can act as adjuvants, enhancing the immune response.
- Dose and Dosing Regimen: Both low and very high doses can impact ADA development. Low doses may not reach the threshold for T-cell activation, while very high doses can sometimes induce immune tolerance. Dosing frequency also plays a role, with higher-frequency regimens sometimes leading to the activation of regulatory T-cells (Tregs), thereby reducing immunogenicity.
- Target and Mechanism of Action (MoA): Drugs targeting immune cells (e.g., those binding to CD40 or CD25 on APCs) or designed to enhance an immune response (e.g., T-cell engagers) carry a higher intrinsic risk of immunogenicity.
The Impact of ADAs on Pharmacokinetics (PK) and ADME
The formation of ADAs can significantly alter a drug's Absorption, Distribution, Metabolism, and Excretion (ADME) profile, complicating both its therapeutic effect and its bioanalysis.
Impact on Bioanalysis: When ADAs are present, the drug exists in two forms: free drug and drug bound in an immune complex (IC). It is critical to understand whether an analytical assay measures free drug, total drug (free + bound), or a combination, as this can drastically affect the interpretation of PK data.
Accelerated or Delayed Clearance:
For large-molecule drugs like monoclonal antibodies (>70 kDa), the formation of ADAs typically accelerates clearance. Large immune complexes are rapidly cleared by phagocytic cells in the liver and spleen, bypassing the protective FcRn recycling pathway. This accelerated clearance can dramatically reduce drug exposure and efficacy, even if the ADAs are non-neutralizing.
For smaller protein drugs (<70 kDa) that are normally cleared by the kidneys, ADA binding increases their size, preventing renal filtration. This can paradoxically prolong the drug's half-life and circulation time.
Altered Distribution: The formation of immune complexes can limit the ability of smaller protein drugs to penetrate tissues, while potentially increasing their accumulation and clearance in the liver.
Strategies for Assessing Immunogenicity
1. The Standard Tiered Approach for ADA Detection
Clinical and preclinical samples are typically evaluated for ADAs using a tiered testing strategy to ensure accuracy and manage throughput:
- Screening Assay: A highly sensitive assay designed to identify all potentially positive samples, minimizing the risk of false negatives.
- Confirmatory Assay: Used to confirm the specificity of the response in screening-positive samples, thereby eliminating false positives.
- Characterization Assays: Confirmed positive samples are further characterized to determine the ADA titer (a measure of magnitude) and, most importantly, to assess their neutralizing activity (NAb).
2. Proactive Immunomodulatory Assessment with Prisys Biotech
While detecting ADAs is crucial, a forward-thinking strategy involves proactively assessing a drug candidate's potential to modulate the immune system before ADAs are even generated. This is where specialized preclinical models become invaluable.
Prisys Biotech offers the T-cell-Dependent Antibody Response (TDAR) assay, a gold-standard model for evaluating a compound's effect on overall immune competence. Key features of our TDAR model include:
- Gold-Standard Evaluation: The TDAR assay is widely recognized as the benchmark for assessing the potential immunomodulatory effects of a drug candidate.
- Robust Induction: The immune response is reliably induced using Keyhole Limpet Hemocyanin (KLH), a potent immunogen.
- Comprehensive Readouts: The assay measures the entire continuum of the immune response, from antigen uptake and presentation to T-cell help, B-cell activation, and final antibody production. Key endpoints include anti-KLH IgM and anti-KLH IgG levels.

By utilizing the TDAR model in non-human primates (NHPs), our clients can gain early insights into whether their drug candidate might suppress or enhance the immune system, providing critical data to inform risk assessment and clinical development strategies.
Immunogenicity is a multifaceted challenge that requires a deep understanding of immunology and robust analytical strategies. Effectively navigating this landscape is essential for the successful development of any biotherapeutic.
Prisys Biotech provides the expertise and advanced preclinical models required to comprehensively evaluate immunogenicity risk. From standard ADA detection to sophisticated immunomodulatory assessments like the TDAR assay in NHPs, we equip our partners with the critical data needed to make informed decisions, mitigate risks, and confidently advance their most promising candidates to the clinic.
Contact us today to learn how Prisys Biotech can support your immunogenicity assessment strategy and accelerate your biologic drug development program.