The high attrition rate in central nervous system (CNS) drug development remains one of the most persistent challenges in modern biomedical research. Despite decades of investment and scientific progress, fewer than 10% of CNS drug candidates entering Phase I trials eventually reach the market. This extraordinarily low success rate highlights a fundamental issue: a persistent translational gap between preclinical models and human neurological diseases.

This article examines the scientific basis of this gap and discusses why nonhuman primate (NHP) models-given their neuroanatomical, physiological, and behavioral proximity to humans-offer a more predictive platform for evaluating therapeutic mechanisms, pharmacokinetics/pharmacodynamics (PK/PD), and safety. The aim is to provide researchers with a strategic perspective to reduce translational risk in CNS drug development.
1. A Hard Reality: The "Valley of Death" in CNS Drug Development
CNS therapeutics consistently show one of the lowest approval rates across all therapeutic areas. Numerous analyses indicate that less than 10% of CNS candidates entering first-in-human trials ultimately obtain regulatory approval.
This failure often occurs despite promising efficacy and safety signals in standard rodent models. The consequences are substantial-multi-billion-dollar development costs, long project timelines, and the inability to achieve expected clinical endpoints.
Such outcomes force a critical question:
Do our existing preclinical systems sufficiently capture the complexity of human neurological disorders?
Increasing evidence suggests that current models, particularly rodent-based systems, frequently fall short when predicting human responses.
2. Root Cause of the Translational Gap: Inherent Limitations of Rodent Models
Rodent models remain indispensable for early discovery due to their genetic manipulability, cost-effectiveness, and short experimental cycles. However, when research progresses to evaluating human-like physiology, complex behavior, and long-term disease progression, several intrinsic limitations become evident.
2.1 Neuroanatomical and Circuit-Level Differences
Human and nonhuman primate brains differ fundamentally from rodent brains in size, cortical organization, and connectivity:
The prefrontal cortex, essential for executive function, cognition, and decision-making, is disproportionately larger and structurally more complex in primates than in rodents.
Rodent cortices lack the gyrencephalic folding and high-level functional specialization found in primates.
Consequently, a therapeutic target validated in simplified rodent circuits may not translate within the redundant, highly specialized neural networks of the human brain.
2.2 Species-Specific Pathophysiology of Neurodegenerative Diseases
Many hallmark pathological features of human diseases cannot be fully reproduced in rodents:
- Tau phosphorylation patterns and the structural forms of pathological tau in humans are not naturally exhibited in rodents.
- Aβ oligomer diversity, aggregation pathways, and plaque maturation differ significantly across species.
Transgenic rodent models often mimic isolated pathological events, rather than the progressive, multifactorial disease trajectories observed in humans.
These discrepancies limit the predictive relevance of rodent models for Alzheimer's disease, Parkinson's disease, frontotemporal dementia, and other neurodegenerative disorders.
2.3 Blood–Brain Barrier (BBB) Differences Affecting Drug Distribution
Accurate prediction of CNS drug exposure requires understanding BBB transporter expression and function. Key differences include:
- Cross-species variability in active efflux transporters such as P-gp and BCRP
- Differences in receptor-mediated transcytosis pathways
- Divergence in endothelial tight junction organization
As a result, brain/plasma ratios in rodents frequently do not extrapolate to humans.
2.4 Behavioral and Cognitive Endpoint Non-equivalence
Rodent behavioral assays often simplify cognitive, emotional, or social functions that are far more complex in humans. Tasks such as maze navigation or basic operant conditioning cannot capture:
- episodic memory
- goal-directed planning
- social cognition
- higher-order emotional regulation
This lack of behavioral homology limits the translational value of rodent efficacy endpoints.
3. Why NHP Models Offer Higher Predictive Power for CNS Research
Nonhuman primates, particularly Macaca fascicularis (cynomolgus monkeys), share close homology with humans in neuroanatomy, neurotransmitter systems, cortical development, immune responses, and lifespan. These similarities enable more accurate assessment of:
- drug penetration across the BBB
- target engagement and downstream pathway effects
- long-term disease progression and chronic dosing
- higher-order behavioral and cognitive outcomes
- imaging biomarkers through MRI, PET, and CT
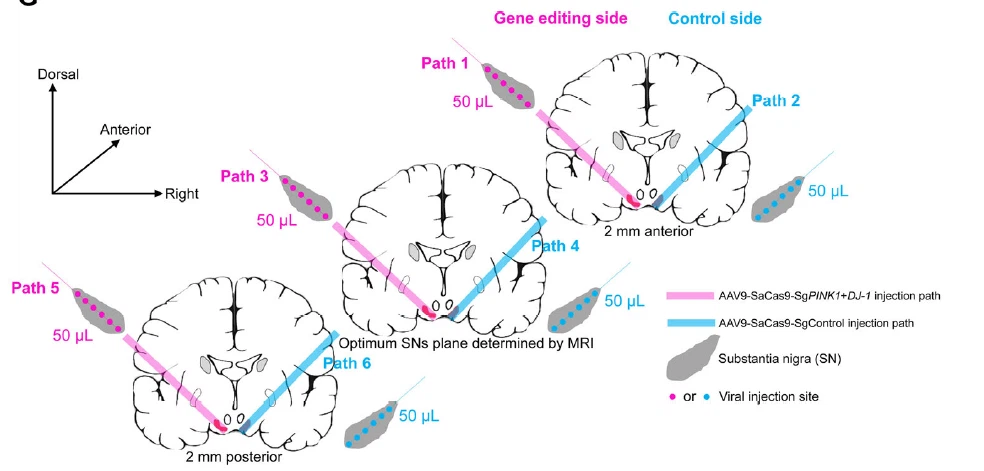
NHP models have shown specific advantages in studies related to:
- Alzheimer's disease (AD): natural age-related amyloid deposition and primate-specific tau biology
- Parkinson's disease (PD): MPTP-induced pathology closely reproducing human nigrostriatal degeneration
- Stroke: primate vascular anatomy and collateral circulation more closely aligned with human cerebrovascular systems
- Gene therapy and biologics: species-matched immune responses and PK/PD dynamics
These attributes make NHPs one of the most reliable platforms for bridging preclinical findings to human clinical outcomes.
4. Toward a More Predictive Preclinical Strategy
Reducing CNS drug attrition requires:
- Refining early-stage discovery with mechanistically relevant models
- Incorporating NHP studies where complex physiology or higher-order function is critical
- Integrating imaging biomarkers (MRI, PET, CT) to enable longitudinal monitoring
- Using NHP PK/PD and biodistribution data to guide dose selection and clinical trial design
By improving preclinical predictivity, researchers can significantly reduce the translational risk associated with first-in-human CNS studies.